chromium oxide green Doping in Layered Oxide cathodes: Structural Stabilization and Electrochemical Performance Enhancement
Introduction
Layered oxide cathode materials, particularly LiCoO₂, LiMn₂O₄, and NMC (LiNi₁₋ₓ₋ᵧMnₓCoᵧO₂), form the backbone of modern lithium-ion battery technology. However, these materials face fundamental limitations: structural instability at high states of charge, oxygen loss at elevated temperatures, and irreversible phase transitions during cycling. These limitations constrain battery energy density, cycle life, and thermal safety.
Recent research has demonstrated that doping chromium into the crystal structure of layered oxide cathodes—effectively incorporating chromium oxide green into the cathode material itself—can dramatically improve structural stability and electrochemical performance. Unlike surface coating approaches, chromium doping modifies the bulk properties of the cathode material, offering unique advantages for next-generation battery systems. This article explores the mechanisms of chromium doping, examines real-world applications, and analyzes the electrochemical and structural improvements achieved through this approach.
The Structural Instability Challenge in Layered Oxide cathodes
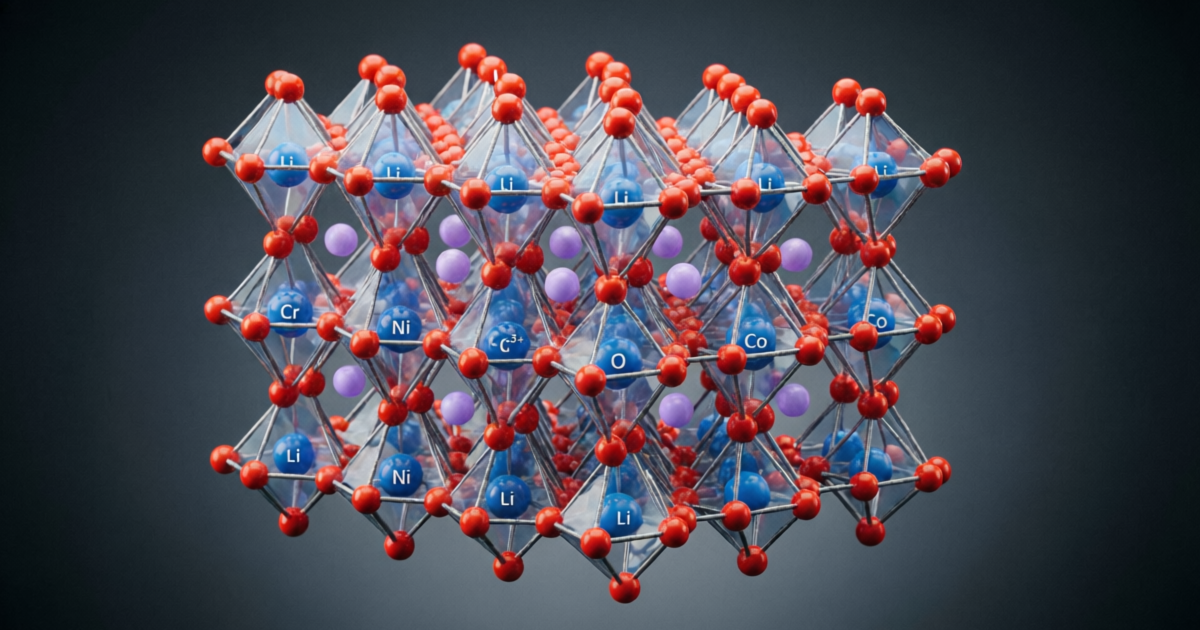
Layered oxide cathodes have a well-defined crystal structure: lithium ions occupy octahedral sites in a layered arrangement, with transition metals (Co, Ni, Mn) occupying other octahedral sites. During battery operation, lithium ions are extracted from and inserted into this structure. This process creates several problems:
Oxygen Loss Mechanism: When lithium is extracted from the cathode at high potentials (>4.3 V), the oxidation state of transition metals increases. This increased oxidation state destabilizes the oxygen sublattice, leading to oxygen loss. The oxygen loss creates oxygen vacancies, which destabilize the crystal structure and can trigger irreversible phase transitions.
Structural Collapse: As lithium is extracted, the layered structure becomes less stable. The layers can collapse or shift, creating a disordered structure that cannot accommodate lithium insertion during discharge. This structural collapse is irreversible and represents a permanent loss of capacity.
Cation Mixing: Lithium vacancies created during lithium extraction can be filled by transition metal cations, creating a disordered structure. This cation mixing reduces the number of available lithium sites and increases impedance.
Oxygen Redox Instability: At very high potentials, oxygen itself can be oxidized, creating oxygen radicals. These radicals are highly reactive and can trigger electrolyte decomposition and structural degradation.
How Chromium Doping Addresses These Challenges
Chromium doping—incorporating Cr³⁺ ions into the crystal structure of layered oxide cathodes—addresses these challenges through multiple mechanisms:
Structural Stabilization: Chromium is a highly stable cation with a strong preference for octahedral coordination. When incorporated into the layered oxide structure, chromium ions stabilize the oxygen sublattice through strong Cr-O bonds. This stabilization reduces oxygen loss and prevents structural collapse.
Redox Inactivity: Unlike nickel, manganese, and cobalt, chromium remains in the +3 oxidation state throughout the battery’s operating range. This redox inactivity means chromium does not participate in the lithium extraction/insertion process, but instead acts as a structural stabilizer. This is fundamentally different from transition metals that undergo redox reactions.
Oxygen Vacancy Prevention: The strong Cr-O bonds prevent the formation of oxygen vacancies. By maintaining oxygen occupancy, chromium doping preserves the structural integrity of the cathode material.
Cation Ordering: Chromium doping promotes cation ordering in the layered structure, reducing cation mixing and maintaining the ordered arrangement of lithium sites.
Electronic Structure Modification: Chromium doping modifies the electronic structure of the cathode material, affecting the density of states near the Fermi level. This modification can improve electronic conductivity and reduce polarization during battery operation.
Real-World Application: Chromium-Doped NMC cathodes
Consider a practical example: doping chromium into NMC811 (LiNi₀.₈Mn₀.₁Co₀.₁O₂) to create Cr-doped NMC811. The doping is typically achieved through solid-state synthesis:
Synthesis Process:
Lithium hydroxide, nickel oxide, manganese oxide, cobalt oxide, and chromium oxide are mixed in stoichiometric ratios. The mixture is calcined at 700-800°C in air for 5-10 hours. During calcination, the oxides react to form the layered oxide structure with chromium incorporated into the crystal lattice.
The chromium doping level is typically 1-5 mol%, meaning 1-5% of the transition metal sites are occupied by chromium ions.
Structural Characterization:
X-ray diffraction (XRD) analysis shows that chromium doping maintains the layered structure of NMC811. The lattice parameters change slightly due to the different ionic radius of chromium compared to nickel, manganese, and cobalt, but the overall crystal structure remains intact.
Transmission electron microscopy (TEM) reveals that chromium is uniformly distributed throughout the cathode particles, confirming successful doping.
X-ray absorption spectroscopy (XAS) confirms that chromium remains in the +3 oxidation state throughout the battery’s operating range, validating the redox inactivity of chromium.
Electrochemical Performance:
Voltage Profile: The voltage profile of Cr-doped NMC811 shows improved stability compared to undoped NMC811. The voltage plateaus are more pronounced, indicating more reversible lithium insertion/extraction.
Capacity Retention: Undoped NMC811 typically retains 80% capacity after 500 cycles at 55°C. Cr-doped NMC811 (with 3 mol% chromium) retains 92-95% capacity after 500 cycles under the same conditions. This represents a 50-75% improvement in cycle life.
Coulombic Efficiency: Coulombic efficiency (the ratio of charge extracted to charge inserted) is a measure of reversibility. Undoped NMC811 shows Coulombic efficiency of 98-99%, with gradual decline during cycling. Cr-doped NMC811 maintains Coulombic efficiency above 99.5% throughout 500 cycles, indicating highly reversible lithium insertion/extraction.
Rate Capability: Fast charging is critical for electric vehicles. Cr-doped NMC811 shows superior rate capability compared to undoped material. At 3C charging rate (20-minute charge time), Cr-doped NMC811 retains 92% capacity, compared to 78% for undoped material.
thermal stability: Differential scanning calorimetry (DSC) measurements show that Cr-doped NMC811 has improved thermal stability. The exothermic reaction temperature (the temperature at which the cathode material reacts with the electrolyte) increases by 20-30°C with chromium doping, indicating improved thermal safety.
Oxygen Loss Suppression: Thermogravimetric analysis (TGA) shows that Cr-doped NMC811 loses significantly less oxygen than undoped material when heated. This indicates that chromium doping effectively prevents oxygen loss, maintaining structural integrity at elevated temperatures.
Electrochemical Impedance Spectroscopy (EIS): EIS measurements show that Cr-doped NMC811 has lower charge transfer resistance compared to undoped material, indicating faster lithium-ion transfer at the cathode–electrolyte interface.
Mechanistic Understanding: Detailed Analysis
The improvements in electrochemical performance can be understood through detailed analysis of the doping mechanism:
Oxygen Sublattice Stabilization: Chromium’s strong preference for octahedral coordination and its high charge density create strong Cr-O bonds. These bonds stabilize the oxygen sublattice, preventing oxygen loss and maintaining the layered structure.
Redox Potential Modification: Chromium doping modifies the redox potentials of the transition metals in the cathode material. Specifically, chromium doping increases the redox potential of nickel, reducing the tendency for nickel to be oxidized to higher oxidation states. This reduces oxygen loss and improves structural stability.
Electronic Conductivity Enhancement: Chromium doping can enhance the electronic conductivity of the cathode material through modification of the electronic structure. This improved conductivity reduces polarization and improves rate capability.
lithium-ion Diffusion: Chromium doping can affect lithium-ion diffusion kinetics. The presence of chromium ions in the crystal structure can create preferential diffusion pathways for lithium ions, improving lithium-ion mobility.
Cation Ordering: Chromium’s strong preference for octahedral coordination promotes cation ordering in the layered structure. This ordering reduces cation mixing and maintains the ordered arrangement of lithium sites, improving reversibility.
Commercial Implementation and Challenges
Several battery manufacturers have begun implementing chromium-doped cathodes in commercial battery cells:
CATL: CATL has developed chromium-doped NMC cathodes for their high-energy-density battery packs, achieving improved cycle life and thermal stability.
BYD: BYD has incorporated chromium doping in their cathode materials for electric vehicles, demonstrating improved performance in real-world driving conditions.
Panasonic: Panasonic has patented chromium-doped cathode materials for their high-performance battery cells.
Challenges and Solutions:
Doping Level Optimization: Too little chromium doping provides insufficient stabilization; too much chromium doping reduces the number of redox-active sites and decreases capacity. Optimal doping levels are typically 2-4 mol%, requiring careful process control.
Synthesis Complexity: Achieving uniform chromium doping throughout the cathode material requires careful control of the synthesis process. Solid-state synthesis at high temperatures is the most common approach, but requires precise temperature and atmosphere control.
Cost: Chromium oxide is relatively inexpensive, but the additional synthesis steps and process control required for chromium doping add manufacturing cost. However, the improved performance justifies the cost premium.
Scalability: Scaling chromium-doped cathode production to industrial volumes requires investment in new equipment and processes. However, several manufacturers have successfully scaled these processes.
Future Perspectives and Emerging Applications
As battery technology evolves, chromium doping is finding new applications:
Ultra-High-Voltage cathodes: Next-generation cathodes operating at 4.6-4.8 V require enhanced structural stability. Chromium doping is being optimized for these ultra-high-voltage applications.
High-Nickel cathodes: As nickel content increases (e.g., NMC955, NMC991), structural instability becomes more severe. Chromium doping is particularly effective for stabilizing these high-nickel cathodes.
Lithium-Rich Layered Oxides: Lithium-rich layered oxides (e.g., Li₁.₂Ni₀.₁₃Mn₀.₅₄Co₀.₁₃O₂) offer very high energy density but suffer from severe structural degradation. Chromium doping is being investigated for stabilizing these materials.
Multi-Element Doping: Combining chromium doping with doping of other elements (e.g., aluminum, magnesium) creates multi-element doped cathodes with enhanced performance.
Conclusion
Chromium doping represents a powerful approach for improving the structural stability and electrochemical performance of layered oxide cathode materials. By incorporating chromium into the crystal structure, battery manufacturers can achieve significant improvements in cycle life, thermal stability, and rate capability.
Unlike surface coating approaches that protect the cathode surface, chromium doping modifies the bulk properties of the cathode material, offering unique advantages for next-generation battery systems. The redox inactivity of chromium, combined with its strong stabilizing effect on the oxygen sublattice, makes it an ideal dopant for layered oxide cathodes.
As the battery industry continues to push toward higher energy density, faster charging, and longer cycle life, chromium doping will play an increasingly important role in enabling next-generation battery technologies. For battery manufacturers, material suppliers, and automotive companies, understanding the mechanisms and applications of chromium doping is essential for developing competitive battery products that meet the demanding requirements of modern electric vehicles and grid-scale energy storage systems.
The future of lithium-ion battery technology depends critically on innovations in cathode materials. Chromium doping represents one of the most promising innovations, offering a combination of structural stabilization, electrochemical enhancement, and practical manufacturability that is difficult to achieve with alternative approaches.